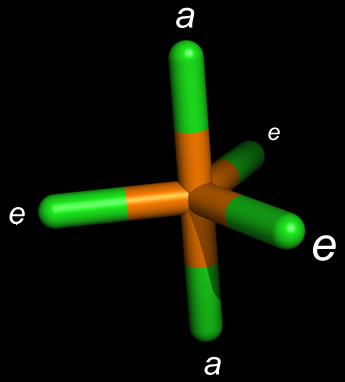
You can see the adjacent bond angles are all in an ideal octahedral geometry, because that is the largest angle that the atoms can be separated while still making six identical #"P"-"F"# bonds. These overlaps generate the #"P"-"F"# bonds. Then, what you will have is six sets of #stackrel("P")overbrace(sp^3d^2)-stackrel("F")overbrace(p_y)# orbital overlaps. The Lewis structure of PF 3 is: Lets count the areas around the phosphorus atom that. PF 3 has a trigonal pyramidal molecular geometry. To make the bonds more uniform, hybridization must occur between the #3s#, #3p#, and #3d# orbitals of phosphorus to generate #sp^3d^2# orbitals. Answer and Explanation: Become a member to unlock this answer Create your account. Otherwise, the bond lengths will not all be the same, when they should be. The problem is that they should be the same bonds for a molecule making six identical #"P"-"F"# bonds. They are both head-on (sigma/ #sigma#) overlaps, but they are not the same bonds. (The ideal bond angles are a separate phenomenon based on electron repulsions.)įor example, an overlap between two #2p_z# orbitals is not the same as the overlap between a #2p_z# and a #3d_(z^2)# orbital, even though they are both possible. Orbital hybridization in #"PF"_6^(-)# requires that all six #"P"-"F"# bonds are identical not necessarily in bond angle, per se, but in the orbitals used to construct the bond. You can see the final shape of this at the bottom. 
This generates an octahedral molecular and electron geometry. That means it can use its #3d# orbitals in addition to its typical #3s# and #3p# valence orbitals. Since phosphorus ( #"P"#, atomic number #15#) is on the third period of the periodic table, it has access to orbitals of principal quantum number #n = \mathbf(3)#. The angle between the fluorine atoms located in axial and equatorial position is 90. The bond angles for the Fluorine atoms in the equatorial position, F-P-F is 120. The molecular geometry, on the other hand, is Trigonal Pyramidal. As mentioned earlier, the fluorine atoms in PF5 either occupy the equatorial position or axial one there are two bond angles for this molecule. You can count these up and see that the number of orbitals used in the hybridization equal the number of electron groups around the central atom. The electron geometry (Electronic Domain Geometry) for PF3 is tetrahedral. We will use valence shell electron pair repulsion (VSEPR) theory to determine its molecular geometry. 
The hybridization of phosphorous in PF 5 is sp 3 d. Phosphorus Trichloride: Phosphorus trifluoride is the name of PF 3.It's a gas that is known for its toxicity. The electron geometry of PF 5 is also Trigonal bipyramidal. In the PF 5 Lewis dot structure, a total of 15 lone pairs and 5 bond pairs are present. 6 electron groups #=>#sp^3d^2# hybridization (EX: #"SF"_6#) The molecular geometry or shape of PF 5 is a Trigonal bipyramidal.5 electron groups #=>#sp^3d# hybridization (EX: #"PF"_5#).4 electron groups #=>#sp^3# hybridization (EX: #"CH"_4#).3 electron groups #=>#sp^2# hybridization (EX: #"BF"_3#).2 electron groups #=>#sp# hybridization (EX: #"CO"_2#).Diatomic molecule #=># No hybridization at all! (EX: #"HCl"#).In general, the typical pattern for basic/simple molecules is:
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
